2020 Volume 3 Issue 3
|
|
INEOS OPEN, 2020, 3 (3), 112–117 Journal of Nesmeyanov Institute of Organoelement Compounds Download PDF
|
|
Polyphenylsilsesquioxane Nanogels as Regulators of the Mechanical Properties of Vulcanizates Based on PDMS
Nesmeyanov Institute of Organoelement Compounds, Russian Academy of Sciences, ul. Vavilova 28, Moscow, 119991 Russia
Corresponding author: M. N. Temnikov, e-mail: temnikov88@gmail.com
Received 4 June 2020; accepted 11 June 2020
Abstract
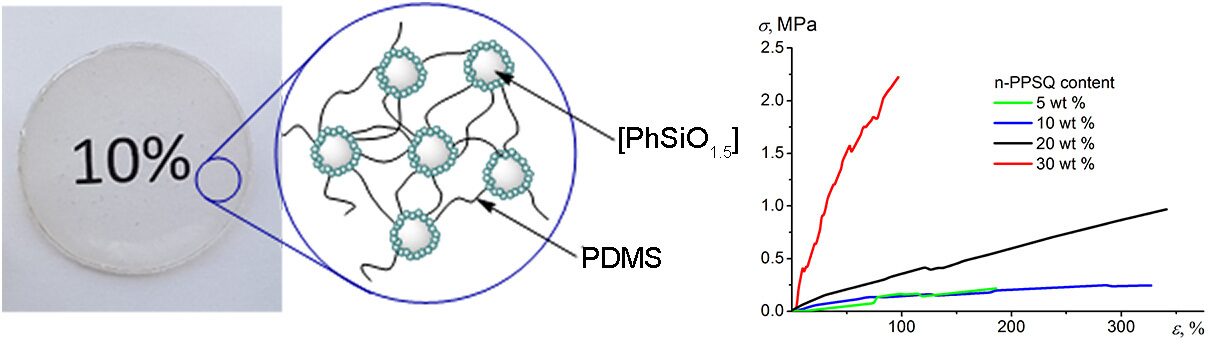
A nanogel featuring a polyphenylsilsesquioxane core and a functional dimethylvinylsilyl shell (n-PPSQ) is used to obtain vulcanizates with polydimethylsiloxane (PDMS) by hydrosilylation. The resulting vulcanizates represent transparent films. The content of n-PPSQ ranges from 5 to 30 wt %. All the vulcanizates exhibit high thermal stability in the air (up to 395 °С). The mechanical characteristics of the films strongly depend on the PPSQ content. On passing from 5 to 30 wt % content of the nanogel, the elastic modulus increases in 60 times from 0.06 to 3.6 MPa. The breaking stress changes from 0.2 to 2.2 MPa.
Key words: polyphenylsilsesquioxane, polydimethylsiloxane (PDMS), block copolymer, mechanical properties.
Introduction
Polyphenylsilsesquioxanes (PPSQs) are one of the most important representatives of organosilsesquioxanes. The practical value of these compounds stems from their properties. As a rule, statistical PPSQs are used as protecting coatings. These coatings display high radiation, thermal and thermooxidative stability, hydrophobicity, and mechanical strength [1–5]. Upon curing, PPSQ resins form transparent films with high refractive indices and excellent dielectric properties [6–8]. The complex of these properties enables the use of PPSQs as electrical insulating thermally stable coatings. The latter ensure the functioning of different devices for prolonged periods of time at 250–300 °С and high humidity [9].
PPSQs are obtained by the hydrolytic condensation of a trifunctional phenylsilane. The choice of the synthesis conditions defines the final product structure and, consequently, its properties. The variation of the initial monomer type [10], its concentration [11], medium pH, catalyst type, surfactant additive [12], and other conditions can afford in the limiting cases PPSQs of polycyclic structures—ladder or polyhedral.
Thus, the anionic polymerization of the hydrolysis products of phenyltrichlorosilane at 250 °С results in the ladder polymers with high molar masses [13]. Harsh synthesis conditions give rise to defects in the structure of a ladder polymer [14]. The control of the amount of these defects, which can affect the product properties, is very challenging. On the other hand, the production of defect-free ladder polymers is more complicated, and the molar masses of the resulting polymers are significantly lower.
The hydrolysis of phenyltrialkoxysilane in a diluted medium leads to the precipitation of the polyhedral PPSQ octamer T8 [15]. The catalyst modification can open the way to other polyhedral PPSQs—Т10 and Т12 [11, 16]. Although the structures of these compounds do not cause doubts, their practical value is not high. This is associated with their low solubility and compatibility with other organosilicon and organic polymers. To address this issue, these PPSQs are modified by the introduction of new functional groups [15].
Harreld et al. [12] suggested the method for controlling the properties of PPSQ by the synthesis conditions. Although the resulting dependences are a step towards the directed modification, the structures of the products are difficult for interpretation. This means that the optimization of the properties has an empirical character.
The understanding of structure–property relationships is multiply complicated on passing to more complex chemical compositions. This problem is particularly relevant for PPSQs. This is connected with the fact that the highest practical value is characteristic of PPSQ copolymers. Indeed, most of commercial polishes based on PPSQs are derived by cohydrolysis and condensation of phenyltrichlorosilane and a bifunctional diorganodichlorosilane [9, 17] since the mechanical properties of the polymer comprised entirely of [PhSiO1.5] structural units have only limited use. The introduction of bifunctional units reduces the brittleness of products and simplifies their processing and application to surfaces.
Although the empirical method is not likely to be eradicated, a transition to the high-tech era requires a principally new level of comprehension of the relations between molecular structures and properties of modern materials.
In this respect, of paramount interest are block copolymers of PPSQ and PDMS. The valuable properties of these copolymers induced their commercial production. Nowadays, these copolymers are produced under a trademark Lestosil. A combination of flexible PDMS blocks with rigid PPSQ blocks endows the products with high strength, hardness, and elasticity [18, 19]. Lestosil copolymers are obtained by the cohydrolysis of PDMS with terminal dimethylchlorosilyl units and phenyltrichlorosilane. Although the properties of these products strongly differ from those of neat PPSQ and its copolymers, there are no unambiguous structures. The problem is that the combined hydrolysis gives rise, along with the block copolymers, to the individual compounds of PPSQ having complex compositions.
An attempt to address this issue was made at INEOS RAS. As a result, an analog of Lestosil, namely, Bloksil was developed [20]. Unlike Lestosil, Bloksil was prepared from PDMS bearing OSiPhCl2 terminal groups. Close natures of the terminal groups and phenyltrichlorosilane afforded a block copolymer with a more uniform structure [21]. However, the structure of the PPSQ block, its molar mass and molar mass distribution are difficult to determine.
Herein, we report on the vulcanizates of PDMS with terminal OSiMe2H groups (PDMS-H) and PPSQ nanogel (n-PPSQ) bearing terminal dimethylvinylsilyl groups. A peculiarity of our approach is the assurance of block structures of the vulcanizates based on PPSQ and PDMS. The sizes of n-PPSQ are determined by the conditions of its synthesis which precedes the vulcanization. This allows one to avoid the step of cohydrolysis which, as it was already shown, leads to products with complex structures and compositions.
Results and discussion
Production of n-PPSQ and the vulcanizates on its base
The main idea of this work consisted in the preliminary production of the PPSQ block and its subsequent reaction with PDMS-H without risk to cleave siloxane bonds. First of all, this approach provided certain sizes of flexible and rigid blocks in the final product. Secondly, it ensured the absence of low-molecular PPSQs of a ladder or polyhedral type. Both factors are important. The former allowed us to evaluate the effect of the ratio of flexible and rigid blocks. This is impossible during cohydrolysis in the case of Lestosil and Bloksil because it is very difficult to define the precise sizes of the PPSQ blocks. The latter is no less important since the presence of low-molecular PPSQs uncertainly affects the final properties of the product.
n-PPSQ was obtained by the previously developed procedure. It consisted in the intramolecular cyclization of hypercrosslinked polyphenylethoxysiloxane (PPEOS). This approach was described in Refs. [22, 23] and is presented in a general form in Fig. 1. As can be seen, n-PPSQ was obtained by the cascade hydrolytic polycondensation of PPEOS in an excess of acetic acid [24]. This process is intensive during refluxing. The ethoxy groups undergo intra- and intermolecular condensation. In 5 h, the reaction was terminated by the addition of a blocking agent. Then, the mixture was continued to reflux for 24 h. The resulting polymers represented a core–shell system with the PPSQ core and the shell with Me2VinSiO terminal groups.
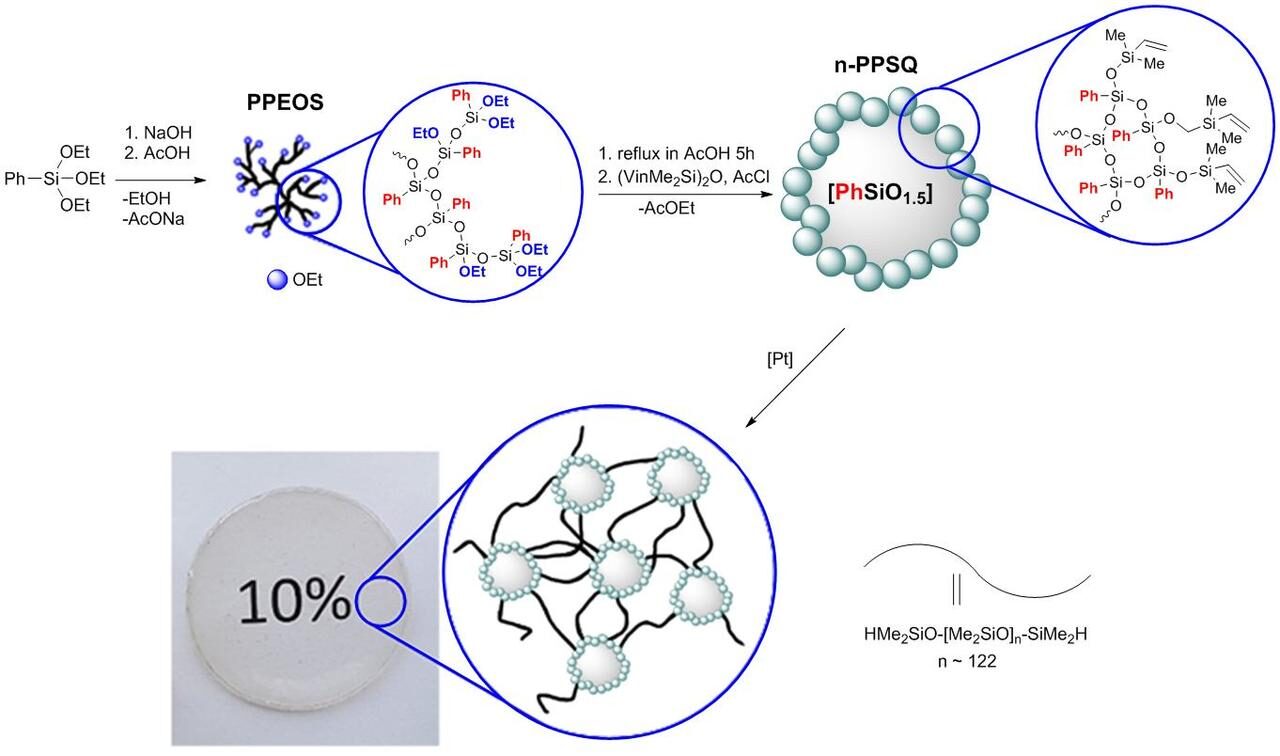
Figure 1. Production of n-PPSQ.
Earlier we showed that the time of condensation in acetic acid defines the density and sizes of the core. An insignificant increase in the molecular weight (MW) leads to considerable changes in the properties. Thus, the dynamic viscosity and glass-transition point (Тg) increased by several orders of magnitude. This was demonstrated by the example of nanogels with the [SiO2] and [MeSiO1.5] core and the shell containing [Me3SiO0.5] terminal groups [25–27]. Recently, we have shown that the same is true for the nanogels with the [PhSiO1.5] core and the shell with [Me3SiO0.5] terminal groups. On passing from the nanogel with MW of 1400 to that with MW of 2560, the dynamic viscosity increased approximately by five orders of magnitude, whereas the value of Tg rose by 110 °С [22]. This is explained by a transition of the character of nanogel flow from macromolecular (segmental) to colloidal (motion of the whole particle).
Hence, the synthesis of n-PPSQ according to the scheme depicted in Fig. 1 furnished the polymer with a globular structure featuring dimethyl(vinyl)silyl functional terminal groups.
The GPC curve and 1Н NMR spectrum of the resulting PPSQ nanogel are presented in Figs. 2 and 3.

Figure 2. GPC curve of the resulting PPSQ nanogel (MW = 8000).

Figure 3. 1Н NMR spectrum of the resulting PPSQ nanogel.
According to the GPC data, the resulting polymer had MW equal to 8000. From the ratio of integral intensities of the signals in the 1Н NMR spectrum, it was obvious that the core sizes considerably exceeded those of the shell. Based on the resulting data, we concluded that the molecular formula of the resulting polymer is [PhSiO1.5]55.2[VinMe2SiO0.5]9.2. However, the value of MW according to the GPC data may be underestimated due to differences in the hydrodynamic radii of a polystyrene coil and n-PPSQ coil. Therefore, the calculated formula should be considered as approximate. At the same time, the ratio of the main and terminal units was right since it was obtained from the 1Н NMR data.
Then, the vulcanizates were obtained by the hydrosilylation over Karstedt's catalyst. The reactions were performed with the polydimethylsiloxane polymer featuring Mn of 9000 (calculated from the 1H NMR spectrum). A solution of the components with the catalyst was poured onto a cellophane substrate and cured for 1 day. This afforded the films bearing 5, 10, 20, and 30 wt % of n-PPSQ calculated relative to the PDMS mass (Fig. 4).

Figure 4. Photographs of the resulting films bearing 5, 10, 20, and 30 wt % of n-PPSQ.
The resulting films at the top of the rings (the upper covers of cylinders) were fully transparent. This is evidenced by the clear captions of the nanogel contents in PDMS.
Thermal properties
TGA studies showed that all the films possess high thermal stability up to 355–395 °С (Fig. 5, Table 1). We anticipated that the thermal stabilities of the resulting vulcanizates would increase proportionally to the contents of n-PPSQ. The comparison of the temperatures of 5% decomposition (Тd5%) in the air (see Table 1) showed that this assumption is correct up to the nanogel content of 20 wt %. At this content of the nanogel, the value of Тd5% of the vulcanizate was higher than that of neat PDMS-H by 20 °С and reached 395 °С. At the nanogel content of 30 wt %, the value of Тd5% reduced to 355 °С, which is already lower than that of PDMS-H by 20 °С. From Table 1 it is obvious that 20 wt % content of n-PPSQ corresponds to the stoichiometric ratio of Si–H and Si–Vin groups. Presumably, a decrease in the thermal stability in the case of 30% filling is associated with the excess of VinMe2SiO groups.

Figure 5. TGA curves of neat PDMS and the resulting films bearing 5, 10, 20, and 30 wt % of n-PPSQ obtained in the air.
Table 1. Properties of the vulcanizates based on PDMS-H and n-PPSQ
|
Content of n-PPSQ, |
E, MPa |
σ, MPa |
ε, % |
Тd5%, °С |
Content of a solid residue, wt % |
Mp, °С |
|
PDMSa |
n/mb |
n/m |
n/m |
375 |
15 |
n/m |
|
5 |
0.06 |
0.2 |
280 |
375 |
21 |
–40.41 |
|
10 |
0.21 |
0.25 |
355 |
380 |
30 |
–42.24 |
|
20 |
0.65 |
1.0 |
380 |
395 |
36 |
–43.25 |
|
30 |
3.6 |
2.2 |
80 |
355 |
57 |
–43.23 |
|
E elastic modulus; σ breaking stress; ε strain-to-fracture; Тd5% temperature of 5% mass loss in the air; Mp melting point; |
On the other hand, vinyl groups can form spatial cross-links by the radical mechanism [28, 29]. The values of solid residues (Fig. 5, Table 1) show that they increase proportionally to the content of the nanogel from 21 to 36%. This corresponds to the filling contents from 5 to 20 wt %, i.e., up to the stoichiometric ratio of the functional groups. At 30% filling, the amount of a solid residue is higher than it could be expected.
The high content of an incombustible solid residue is promising for the potential application of these materials in electrical insulation. During burning, this insulation releases small amounts of toxic compounds. The solid residue continues to protect the wire.
Figure 6 presents the DSC curves of the resulting vulcanizates which clearly show the melting peaks of PDMS. This indirectly confirms that the polymer has a block structure in all the samples. Otherwise, the crystallization would be impossible and we would observe only one (in the case of full overlapping) or two (in the case of partial overlapping) glass-transition peaks.
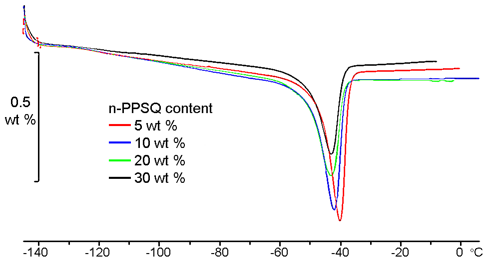
Figure 6. DSC curves of the resulting vulcanizates.
Mechanical properties of the resulting vulcanizates
Most explicitly the effect of the degree of filling with n-PPSQ was reflected on the mechanical properties of the resulting vulcanizates.
An increase in the nanogel content in the composition led to essential changes in the elastic modulus (Table 2). The stress–strain curves (Fig. 7) show that this parameter increases in 60 times on passing from 5 to 30 wt % of n-PPSQ. The breaking stress also significantly increases. The strain-to-fracture grows from 280 to 380% for the vulcanizates bearing 5 and 20 wt % of n-PPSQ, respectively (Table 2).
Table 2. Content of n-PPSQ relative to PDMS-H
|
Content of |
m (PDMS), g |
m (n-PPSQ), g |
SiH/SiCH=CH2 |
|
5 |
1.19 |
0.06 |
3.8 |
|
10 |
1.10 |
0.11 |
2 |
|
20 |
1.01 |
0.20 |
1 |
|
30 |
0.90 |
0.27 |
0.65 |
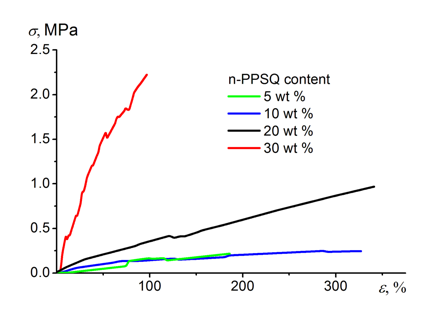
Figure 7. Stress–strain curves of the resulting vulcanizates.
These parameters (σ = 0.65 MPa, ε = 380%) are comparable to those of Elastosil 1110 commercial sealant (σ = 0.8 MPa, ε = 140%) and exceeds those of Elastosil 1102 and Elastosil 2103 (σ = 0.2–0.3 MPa, ε = 100–200%) [9].
A further increase in the content of n-PPSQ to 30 wt % afforded a stronger and less elastic vulcanizate: σ = 2.2 MPa, ε = 80%. This can be explained by the formation of stronger intermolecular interactions compared to the classical cured PDMSs. These interactions take place in phenyl-substituted systems owing to π–π stacking [30]. This means that the nanogel, besides the role of a cross-linking agent, acts as a molecular filler. Nevertheless, the vulcanizate remains transparent (see Fig. 2). This makes such a composition attractive for potential application in optoelectronics. By the mechanical properties, the composition of PDMS-H with 30 wt % of n-PPSQ is comparable to Viksint K-18 commercially available compound (σ = 1.7 MPa, ε = 80%) [9].
When comparing the vulcanizates obtained in this work with the commercial trademarks, one should recall that the latter represent considerably more complex compositions. The sealant and compound trademarks contain, besides a polymer basis, a curing agent, a filler, a plasticizing agent, and a range of other specific additives. This implies that the systems presented in this work have great potential for further modification to strengthen their properties associated with certain practical applications. The tuning tools can be both the molecular structure of a polymer matrix (ratio of the blocks, block sizes) and the introduction of different additives, for example, fillers or plasticizing agents, combination with organic polymers, etc.
Experimental
Materials and methods
All the solvents were purified by the standard procedures [31]. The starting alkoxysilanes were distilled over sodium immediately prior to use. NaOH (Fluka), phenyltriethoxysilane, 1,3-divinyl-1,1,3,3-tetramethyldisiloxane, 1,1,1,3,3,3-hexamethyldisiloxane, Karstedt's catalyst (a solution of Pt(0) complex of 1,3-divinyl-1,1,3,3-tetramethyldisiloxane in xylene with the Pt content of about 2%), and acetyl chloride (ABCR) were purchased from commercial sources. PDMS-Н was obtained by the cationic polymerization of octamethylcyclotetrasiloxane and tetramethyldisiloxane in the presence of KU-23 sulfocationite resin. The resulting sample of PDMS had the molar mass Mn of 9000 (according to the 1Н NMR spectroscopic data). Toluene was purified by refluxing and distillation over calcium hydride. Acetic acid was dried by distillation over phosphorus(V) oxide.
The NMR spectra were registered on a Bruker Avance III HD 400 spectrometer (Germany) operating at 400 MHz for 1H nuclei. Tetramethylsilane was used as an internal reference. CDCl3 was used as a solvent. The spectra were processed with Mestrenova software.
GPC analysis was performed on a Shimadzu instrument (Japan, Germany) with a RID-20A refractometer detector and a Phenogel 1000A column (size (300 × 7.8 mm)); tetrahydrofuran was used as an eluent. The GPC columns were calibrated with polystyrene standards (PSS).
The DSC studies were carried out on a Mettler-822e unit at the heating rate of 10 °С/min in an argon atmosphere.
The thermogravimetric (TGA) analysis was performed on a Derivatograph C unit (MOM, Hungary) using 20 mg samples at the heating rate of 5 °С/min in the air.
The mechanical tests of the films were performed on a Lloyd Instruments LR10KPlus test machine.
Syntheses
Production of PPSQ nanogels with VinMe2SiO terminal groups. The target nanogels were synthesized starting from hypercrosslinked polyphenyl(ethoxy)siloxane (PPEOS) obtained, in turn, by the published procedure [23].
Acetic acid (400 mL) was added to polyphenyl(ethoxy)siloxane (28 g, 0.17 mol). The resulting mixture was refluxed for 5 h. After cooling to room temperature, 1,3-divinyl-1,1,3,3-tetramethyldisiloxane (90 g, 0.48 mol) and acetyl chloride (0.22 g, 0.0028 mol) were added. The reaction mixture was continued to reflux for 24 h, then diluted with toluene (20 mL), washed with water, and dried over anhydrous Na2SO4.
The target product was purified from low-molecular fractions by reprecipitation. For this purpose, the polymer (20 wt % of n-PPSQ) was dissolved in THF, and the resulting solution was poured into an excess of the precipitating agent. The precipitated polymer was decanted and dried at the residual pressure of 0.001 bar. Yield: 74%.
Production of the films based on PDMS and n-PPSQ with dimethylvinylsilyl terminal groups. The curing of the films was carried out on a cellophane substrate applied to a glass ring with a diameter of 72 mm. The reagent amounts were calculated using the following formulae:

where V is the volume of the reagents, S is the substrate area, l is the film thickness, and d is the ring diameter.
n-PPSQ was added in the amount of 5, 10, 20, and 30 wt % relative to PDMS (Table 2).
In all cases, 0.1 μL of a solution of Karstedt's catalyst was added. All the films were cured for 24 h. After separation of the film from the cellophane substrate, it was left under vacuum in a drying oven at 60 °С and the residual pressure of 0.001 bar for 8 h.
Conclusions
We developed an alternative route for the production of block copolymers based on PDMS and PPSQ, which utilizes, unlike the syntheses of Lestosil and Bloksil analogs, polyphenylsilsesquioxane nanogels (PPSQ block) and polydimethylsiloxane telechelics. The vulcanizates were obtained by the hydrosilylation of n-PPSQ and PDMS bearing terminal HMe2SiO groups. The resulting films containing 5, 10, 20, and 30 wt % of n-PPSQ were fully transparent. The DSC studies confirmed indirectly their block structures. The mechanical properties of the resulting vulcanizates were found to strongly depend on the ratio of the reagents. Thus, the elastic modulus increases in 60 times and the breaking stress grows in 11 times on passing from 5 to 30 wt % of n-PPSQ. All the vulcanizates are elastic; their strain-to-fracture comprises 80–380%. The mechanical properties of the resulting vulcanizates are comparable to those of the commercial sealants and compounds, even without the use of conventional fillers for the popular sealants.
Acknowledgements
This work was supported by the Russian Foundation for Basic Research, project no. 18-33-00909.
The NMR spectroscopic studies were performed with the financial support from the Ministry of Science and Higher Education of the Russian Federation using the equipment of the Center for Molecular Composition Studies of INEOS RAS.
References
- A. Provatas, J. G. Matisons, Trends Polym. Sci., 1997, 5, 327–332.
- L. N. Pankratova, L. T. Bugaenko, A. A. Revina, High Energy Chem., 2000, 34, 16–22. DOI: 10.1007/BF02761782
- N. A. Senatova, V. T. Nikolenkov, V. N. Spektor, R. S. Sharafiev, Dokl. Akad. Nauk, 1994, 335, 68–69.
- M. Morita, S. Imamura, A. Tanaka, T. Tamamura, J. Electrochem. Soc., 1984, 131, 2402–2406. DOI: 10.1149/1.2115266
- A. Oikawa, S.-i. Fukuyama, Y. Yoneda, H. Harada, T. Takada, J. Electrochem. Soc., 1990, 137, 3223–3229. DOI: 10.1149/1.2086191
- S. Hayashida, S. Imamura, J. Polym. Sci., Part A: Polym. Chem., 1995, 33, 55–62. DOI: 10.1002/pola.1995.080330106
- JP Patent 04 12.333, 1992.
- JP Patent 06 109.936, 1994.
- M. V. Sobolevskii, Oligoorganosiloxanes: Properties, Production, and Use, Khimiya, Moscow, 1985 [in Russian].
- L. A. S. De A. Prado, E. Radovanovic, H. O. Pastore, I. V. P. Yoshida, I. L. Torriani, J. Polym. Sci., Part A: Polym. Chem., 2000, 38, 1580–1589. DOI: 10.1002/(SICI)1099-0518(20000501)38:9<1580::AID-POLA22>3.0.CO;2-7
- S.-S. Choi, A. S. Lee, S. S. Hwang, K.-Y. Baek, Macromolecules, 2015, 48, 6063–6070. DOI: 10.1021/acs.macromol.5b01539
- J. H. Harreld, K. Su, D. E. Katsoulis, M. Suto, G. D. Stucky, Chem. Mater., 2002, 14, 1174–1182. DOI: 10.1021/cm010695i
- J. F. Brown Jr., L. H. Vogt Jr., A. Katchman, J. W. Eustance, K. M. Kiser, K. W. Krantz, J. Am. Chem. Soc., 1960, 82, 6194–6195. DOI: 10.1021/ja01508a054
- K. A. Andrianov, S. V. Bushin, M. G. Vitovskaya, V. N. Emel'yanov, P. N. Lavrenko, N. N. Makarova, A. M. Muzafarov, V. Ya. Nikolaev, G. F. Kolbina, I. N. Shtennikova, V. N. Tsvetkov, Polym. Sci. U.S.S.R., 1977, 19, 536–543. DOI: 10.1016/0032-3950(77)90107-1
- R. M. Laine, M. F. Roll, Macromolecules, 2011, 44, 1073–1109. DOI: 10.1021/ma102360t
- J. C. Furgal, T. Goodson III, R. M. Laine, Dalton Trans., 2016, 45, 1025–1039. DOI: 10.1039/C5DT04182A
- K. A. Andrianov, L. M. Khananashvili, Technology of Organoelement Monomers and Polymers, Khimiya, Moscow, 1973 [in Russian].
- A. M. Chaikun, M. A. Venediktova, Ya. A. Bryk, Proc. VIAM, 2019, 1, 21–30. DOI: 10.18577/2307-6046-2019-0-1-21-30
- R. M. Minas'yan, A. N. Polivanov, O. I. Minas'yan, Polym. Sci., Ser. D, 2016, 9, 40–42. DOI: 10.1134/S1995421216010123
- Yu. N. Bubnov, K. A. Kochetkov, Chem. Heterocycl. Compd., 2004, 40, 1375–1379. DOI: 10.1007/s10593-005-0090-y
- L. N. Pankratova, V. N. Sokolov, N. V. Mazurina, E. S. Obolonkova, A. Yu. Rabkina, L. I. Kuteinikova, B. G. Zavin, Vysokomol. Soedin., Ser. B, 1997, 39, 1711–1714.
- M. N. Temnikov, V. G. Vasil'ev, M. I. Buzin, A. M. Muzafarov, Eur. Polym. J., 2020, 130, 109676. DOI: 10.1016/j.eurpolymj.2020.109676
- M. N. Temnikov, M. I. Buzin, N. V. Demchenko, G. V. Cherkaev, N. G. Vasilenko, A. M. Muzafarov, Mendeleev Commun., 2016, 26, 121–123. DOI: 10.1016/j.mencom.2016.03.012
- E. V Egorova, N. G. Vasilenko, N. V Demchenko, E. A. Tatarinova, A. M. Muzafarov, Dokl. Chem., 2009, 424, 15–18. DOI: 10.1134/S0012500809010042
- N. V. Voronina, I. B. Meshkov, V. D. Myakushev, T. V. Laptinskaya, V. S. Papkov, M. I. Buzin, M. N. Il'ina, A. N. Ozerin, A. M. Muzafarov, J. Polym. Sci., Part A: Polym. Chem., 2010, 48, 4310–4322. DOI: 10.1002/pola.24219
- A. I. Amirova, O. V. Golub, I. B. Meshkov, D. A. Migulin, A. M. Muzafarov, A. P. Filippov, Int. J. Polym. Anal. Charact., 2015, 20, 268–276. DOI: 10.1080/1023666X.2015.1013185
- A. Ya. Malkin, M. Yu. Polyakova, A. V. Subbotin, I. B. Meshkov, A. V. Bystrova, V. G. Kulichikhin, A. M. Muzafarov, J. Mol. Liq., 2019, 286, 110852. DOI: 10.1016/j.molliq.2019.04.129
- S. Hamdani, C. Longuet, D. Perrin, J.-M. Lopez-cuesta, F. Ganachaud, Polym. Degrad. Stab., 2009, 94, 465–495. DOI: 10.1016/j.polymdegradstab.2008.11.019
- E. Delebecq, S. Hamdani-Devarennes, J. Raeke, J.-M. Lopez Cuesta, F. Ganachaud, ACS Appl. Mater. Interfaces, 2011, 3, 869–880. DOI: 10.1021/am101216y
- Silicon Based Polymers: Advances in Synthesis and Supramolecular Organization, F. Ganachaud, S. Boileau, B. Boury (Eds.), Springer, Dordrecht, 2008. DOI: 10.1007/978-1-4020-8528-4
- W. L. F. Armarego, C. L. L. Chai, Purification of Organic Chemicals, 5th ed., 2003, ch. 4, pp. 80–388. DOI: 10.1016/B978-075067571-0/50008-9





