2019 Volume 2 Issue 4
|
|
INEOS OPEN, 2019, 2 (4), 112–123 Journal of Nesmeyanov Institute of Organoelement Compounds DOI: 10.32931/io1916r |
|
Highly Efficient Wavelength Shifters: Design, Properties, and Applications
Enikolopov Institute of Synthetic Polymeric Materials, Russian Academy of Sciences, ul. Profsoyuznaya 70, Moscow, 117393 Russia
Corresponding author: O. V. Borshchev, e-mail: borshchev@ispm.ru
Received 19 August 2019; accepted 8 October 2019
Abstract
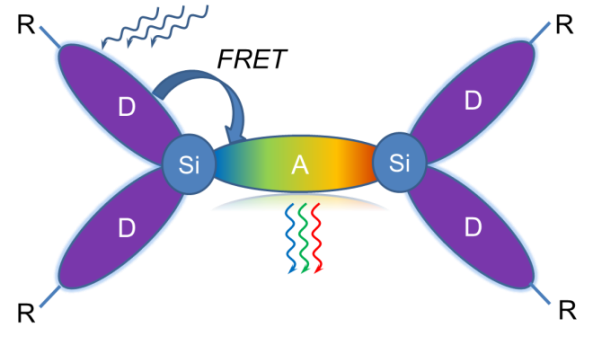
The current review is devoted to efficient wavelength shifters and their applications. The main focus is on the authors' own works that deal with nanostructured organosilicon luminophores (NOLs), which exhibit highly efficient Förster resonance energy transfer (FRET). The examples of devices that utilize these luminophores are provided. The potential paths for future development and prerequisites for successful commercialization of these systems are outlined.
Key words: nanostructured organosilicon luminophores (NOLs), Förster resonance energy transfer (FRET), luminescent downshifting, white LEDs.
1. Introduction
Effective shifting of a light wavelength is an urgent task in many fields of science and technology. Spectral shifters are particularly popular in different optoelectronic devices, which are used in photonics, nuclear physics, and elementary particle physics. Thus, most of detectors of Cherenkov radiation utilize wavelength-shifting devices; this is caused by the low sensitivity of photodetectors in the ultraviolet spectral range [1, 2]. Detectors of low-energy elementary particles based on noble gases include the materials which reemit ultraviolet light to the visible spectrum [3–5]. To improve the efficiency of conversion of sunlight into electrical energy in photovoltaic cells, the spectral characteristics of incident light are modified [6–9]. All the modern illumination systems utilize the reemission of ultraviolet or blue light from a light-emitting diode into green, yellow, or red spectral ranges for production of white light [10–12].
There are three main mechanisms for shifting the light wavelength: cascade luminescence (downconversion), anti-Stokes luminescence (upconversion), and photoluminescence (downshifting). In the case of downconversion, a high-energy photon is divided into lower-energy photons [13]. The downconversion can be realized for vacuum ultraviolet photons (VUV photons), since the energy of these photons is more than double the energy of photons in the near-UV and visible spectral ranges. This phenomenon of two-photon luminescence, which is also called as quantum cutting, quantum splitting, or photon cascade emission, was predicted by Dexter in the 1950s [14, 15] and was demonstrated experimentally 20 years later [16, 17].
In the case of the anti-Stokes luminescence, the photons are summed: two or more low-energy photons afford a photon with the higher energy [18]. This is the way how a long-wave radiation with the low photon energy is transformed into a short-wave radiation which photons have the higher energy. The first reports on the anti-Stokes luminescence were published in 1959. [19, 20]. The anti-Stokes luminophores are used for imaging of the infrared radiation of lasers [21] and optical imaging of tumor tissues [22]. The cascade and anti-Stokes luminescence are usually realized using inorganic chromophores, which luminescence depends on their compositions and crystalline structures. The wavelength shifting by the third mechanism (downshifting) is accomplished generally due to the photoluminescence of organic molecules, which is almost always caused by the π-electron system of a separate molecule [23].
This review is concerned with organic spectral shifters featuring high efficiencies. Particular attention will be drawn to the molecules with intramolecular energy transfer. The potential application fields and the future of these systems will also be discussed.
2. Organic spectral shifters
In general, an efficient spectral shifter can be any organic luminophore that possesses a certain set of properties: a broad absorption cross-section, which is related to the molar absorption coefficient (ε), the highest Stokes shift (S), and a high photoluminescence quantum yield (PLQY). To shift the spectrum of incident light, a luminophore must, first of all, effectively absorb an incident electromagnetic radiation; therefore, its molar absorption coefficient must have the maximum possible value. A Stokes shift is defined as a difference between a maximum of the most long-wave absorption band and a luminescence maximum. The high Stokes shift implies low overlapping between absorption and emission, which leads to the minimum optical losses due to self-absorption, especially at the high luminophore concentration. The efficiency of light reemission is determined by the luminescence quantum yield of the molecule under consideration, i.e., the ratio of the number of photons of the luminescence radiation to the number of absorbed photons of the exciting light. Hence, if an organic luminophore possesses all three mentioned features, it can be defined as an efficient spectral shifter.
A large number of organic luminophores with various chemical structures have been synthesized to date [24]. They include several classes of compounds that can be used as spectral shifters. First of all, these are conjugated aromatic hydrocarbons and their derivatives [1, 25–29] (Fig. 1). In these systems, an increase of the number of aromatic moieties leads to a simultaneous growth of the molar extinction coefficient and a shift of the absorption and emission maxima to the long-wave region, and also to a concomitant exponential decrease of the luminophore solubility in organic solvents and polymer matrices, which is especially important for their practical application [30–33].
Figure 1. Examples of aromatic hydrocarbons.
One of the most popular classes of luminophores, which are used as spectral shifters, are the compounds bearing five- and six-membered heterocycles [34–36] (Fig. 2). The introduction of heterocycles into π-conjugated systems affords intensive luminescence. Most luminophores of this class exhibit luminescence properties both in diluted solutions and in the crystalline state, with the maxima of spectral characteristics from the violet to red color [37–40]. The main drawback of these compounds is the lower photostability compared to aromatic hydrocarbons [41, 42].
Figure 2. Examples of π-conjugated molecules containing heteroatoms.
The last 20 years have witnessed a rapid development of new luminophores based on 4,4-difluoro-4-bora-3a,4a-diaza-s-indacene (BODIPY) [43–45]. They usually feature high extinction coefficients and high luminescence quantum yields. The chemical modification of BODIPY afforded luminophores that emit in a broad spectral range: from blue to red [46–49]. At the same time, the spectral shifters based on BODIPY exhibit low Stokes shifts and are prone to concentration quenching.
2.1. Spectral shifters exhibiting an efficient intramolecular energy transfer
In an attempt to overcome the main shortcomings of classical organic luminophores, we suggested the concept of nanostructured organosilicon luminophores (NOLs) [50–52]. NOLs represent branched or dendritic organosilicon molecular antennae that are able to capture light quanta (photons) and nonradiatevely transfer the electronic excitation energy from the periphery to the nucleus, which leads to effective light absorption in a broad spectral range and emission in a narrow, more long-wave region [53–55]. In NOLs two spectrally different types of chromophores are connected with each other through the silicon atoms, which fix them in space at the distance of 1–2 nm between gravity centers of the chromophores. There is no conjugation between π-electron systems of the chromophores connected through the silicon atom. However, at short distances, a transfer of electronic excitation energy between them can proceed by the Förster mechanism (FRET) [56]. In this case, the whole molecule will take part in absorption, and the energy of electronic excitation of the chromophores with the wider forbidden zone (a donor fragment) will be transferred to a fragment with the narrower forbidden zone (an acceptor fragment) and will be emitted by it [57–59] (Fig. 3).
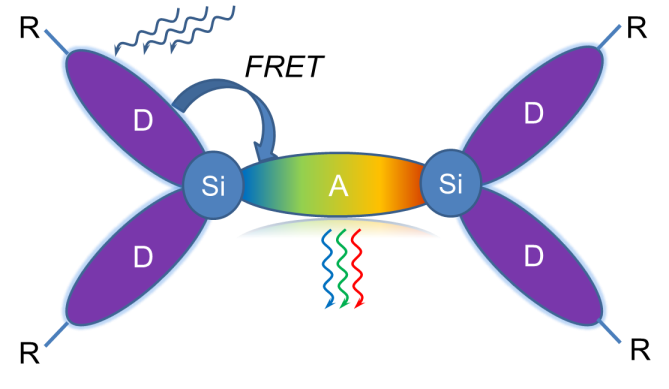
Figure 3. Schematic representation of NOLs.
A probability of the excitation transfer by the Förster mechanism is inversely proportional to the sixth power of the distance (R) between an energy donor (D*) and an acceptor (A*): rate constant (D* → A*) = 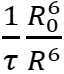 , where R0 is the critical distance for the energy transfer, at which the transfer and the radiative deactivation are equally possible (the Förster radius); τ is the real average lifetime of the excited donor molecule in the absence of an energy transfer. At the fixed distance between the donor and acceptor fragments, the higher R0, the higher the probability of energy transfer by the Förster mechanism. The critical distance of an energy transfer R0 increases with a growth of the photoluminescence quantum yield (PLQY) of the donor, the molar extinction coefficient (ε) of the acceptor, and the overlapping degree of the donor photoluminescence and acceptor absorption spectra. Adherence to the mentioned conditions allows one to reach an efficient intramolecular excitation energy transfer even in the absence of conjugation between the chromophoric fragments of NOLs.
, where R0 is the critical distance for the energy transfer, at which the transfer and the radiative deactivation are equally possible (the Förster radius); τ is the real average lifetime of the excited donor molecule in the absence of an energy transfer. At the fixed distance between the donor and acceptor fragments, the higher R0, the higher the probability of energy transfer by the Förster mechanism. The critical distance of an energy transfer R0 increases with a growth of the photoluminescence quantum yield (PLQY) of the donor, the molar extinction coefficient (ε) of the acceptor, and the overlapping degree of the donor photoluminescence and acceptor absorption spectra. Adherence to the mentioned conditions allows one to reach an efficient intramolecular excitation energy transfer even in the absence of conjugation between the chromophoric fragments of NOLs.
The chemistry of silicon enables the creation of various structures using donor and acceptor moieties of different natures. Figure 4 shows the chemical formulae of some of NOLs obtained. The donor fragments include different conjugated oligomers (p-terphenyl, bithiophene, and РРО) that absorb in the ultraviolet and blue spectral ranges and contain different linear and branched terminal aliphatic groups, which improve the solubility of NOLs in organic solvents and polymer matrices. The choice of a chemical structure of the acceptor moiety is dictated by the required energy of emitted photos and can vary in a very broad range [50, 57, 60].
Figure 4. Examples of NOLs.
The syntheses of NOLs of various chemical structures are carried out using modern methods of organic chemistry [61–63]. The strategy for synthesis of NOLs can be divided into several key steps (Fig. 5). At the first step, functional precursor I with solubilizing group R is synthesized, which either represents the preformed donor moiety (D) or will become a part of the NOL donor fragment. Then, multifunctional branching organosilicon center II is synthesized that can react at the next step with precursor I, resulting in functional monodendrone III.

Figure 5. Schematic representation of the main synthetic steps for the preparation of NOLs [60].
Depending on the amount of reactive groups in a branching center, organosilicon compounds with different contents of donor fragments can be obtained. From the viewpoint of simplicity of molecular design, an optimal structure is a monodendrone with two donor moieties, which can give rise to NOL with four peripheral donor fragments, and one central acceptor moiety (A). In this case, an optimal combination of synthesis simplicity, good reaction yields, and desired optical properties is reached. Although the production of a monodendrone with three donor fragments has certain advantages from the viewpoint of a growth of the molar extinction coefficient, it is associated with the lower yields at the step of the monodendrone synthesis and, consequently, complicates its preparation. At the last step, after several transformations, the resulting functional monodendrone reacts with a bifunctional precursor (B) to give the target NOL structure. After the reaction completion and thorough purification by different methods of organic and polymer chemistry, including recrystallization, high-performance adsorption or gel permeation chromatography, the new nanostructured organosilicon luminophore is isolated in an individual form.
Consider the optical properties of NOLs using the example of NOL16 molecule which has six p-terphenyl donor fragments and a 4,7-diphenyl-2,1,3-benzothiadiazole acceptor moiety. Figure 6 depicts the absorption and luminescence spectra of NOL16.

Figure 6. Absorption and luminescence spectra of NOL16.
Two bands in the absorption spectrum of NOL16 correspond to absorption of the donor (with the maximum at 296 nm) and acceptor (with the maximum at 386 nm) fragments. The profile and maximum of the emission spectrum do not depend on the excitation wavelength. Consequently, there is observed an intramolecular excitation transfer from the NOL16 donor moiety to the acceptor one. The luminescence quantum yield upon excitation at 296 nm is equal to 94±4%, and that upon excitation at 386 nm—99±4%. Therefore, a transfer of the electronic excitation energy from the NOL16 donor fragments to the central acceptor moiety occurs with the high efficiency.
The unique structures of NOLs enable variation of their optical properties over a very broad range. For example, changing the type of donor fragments can lead to a significant modification of the absorption spectrum, leaving the emission spectrum unchanged (Fig. 7). At one and the same acceptor moiety, the application of different chromophores as donors affords NOLs with intensive absorptions in different spectral regions. Thus, the use of substituted p-terphenyl donor moieties leads to the appearance of an intensive absorption band at 262 nm in the spectrum of NOL6. Bithiophene groups afford an intensive band at 328 nm in the absorption spectrum of NOL9, and terthiophene moieties—an intensive band at 378 nm in the spectrum of NOL15. At the same time, the emission spectra of all the luminophores are almost identical and correspond to the luminescence of the acceptor moiety.

Figure 7. Tuning the absorption spectra of NOLs: NOLs with different donor luminophores.
A dendrite structure allows one to essentially extend the absorption cross-section of NOL by increasing the number of donor fragments around a single acceptor moiety. In the case of linear structures, an increase of the number of donor structures relative to that of the acceptor ones leads to a sharp drop of the transfer efficiency and, as a consequence, to a reduction in the quantum yield upon excitation of the donor fragments. This is not observed in dendrite structures. The application of NOLs with the broad absorption cross-section of donor fragments affords wavelength-shifting compositions with the lower self-absorption of luminescence at the same light capture efficiency as in the case of common luminophores [52].
Figure 8 demonstrates the variation of the NOL cross-section with an increase in the number of donor fragments. In the case of an acceptor without donor moieties (n = 0), there is almost no absorption at 336 nm. The extinction coefficient of NOLs with four external fragments (n = 4) composes 93000 M‑1 cm–1; an increase of the number of donors to six (n = 6) leads to a growth of ε at 336 nm to 137000 M–1 cm–1; the branched structure with twelve donor fragments (n = 12) possesses ε equal to 257000 M–1 cm–1. The quantum yield of the acceptor without donor fragments (n = 0) is equal to 88%, and the quantum yields upon excitation of the acceptor fragments of NOLs with n = 4–12 range from 87% to 91%. The ratio of the luminescence quantum yield of NOLs upon excitation of the donor fragments to the quantum yield upon direct excitation of the acceptor moiety changes from 0.92 to 0.98. As can be seen, an increase in the ratio of the number of donor structures to that of the acceptor ones up to 12:1 did not afford an essential reduction in the transfer efficiency.

Figure 8. Absorption (plotted as the molar extinction coefficient distribution; a dotted line) and luminescence (a solid line) spectra of
NOLs with different donor:acceptor ratios (n) [52].
The luminescence spectra of NOLs can readily be tuned for any part of the visible spectral range. For this purpose, different luminophores, which emit in the desired spectral region, are used as acceptor fragments in structures of NOLs. The absorption spectra of these NOLs comprise a superposition of the absorption spectra of the donor and acceptor fragments with the maximum in the region of absorption of donor fragments (Fig. 9a). Owing to the high efficiency of the excitation energy transfer to the acceptor, their emission spectra correspond to the emission spectrum of the chosen luminophore (Fig. 9b).
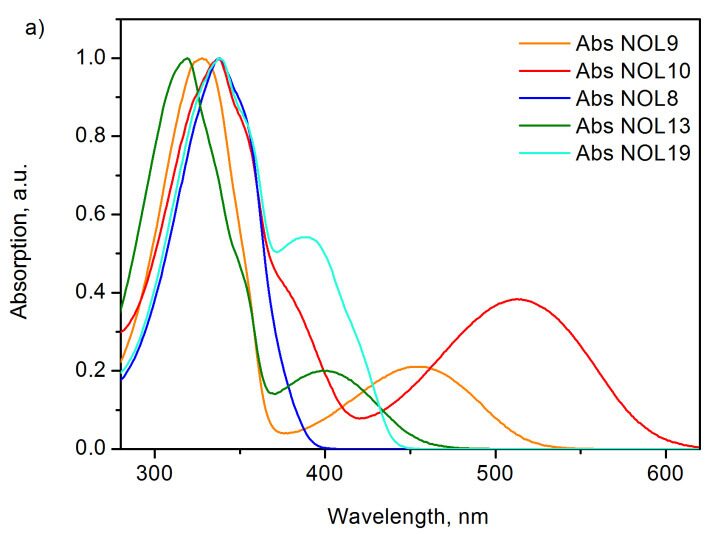
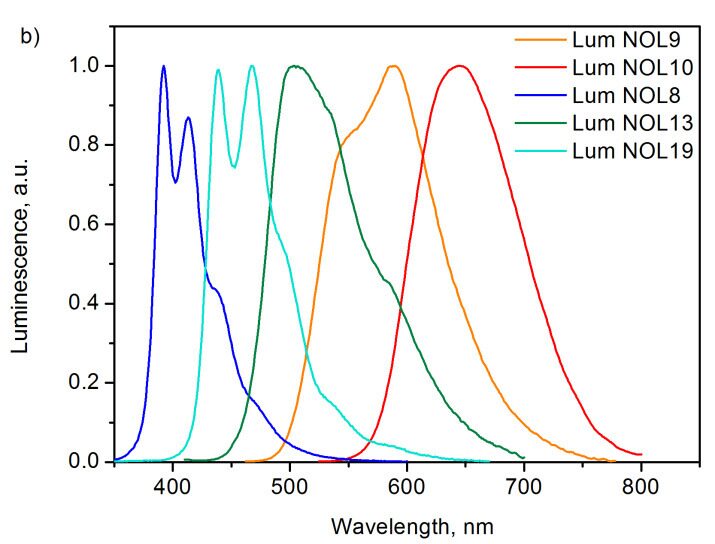
Figure 9. Tuning the luminescence spectra of NOLs bearing the same donors, but different acceptor luminophores:
(a) absorption spectra, (b) luminescence spectra.
Nowadays, a catalog of LumInnoTech LLC [64], which is a holder of patents on the synthesis of this class of luminophores [65–70], includes over 50 NOLs, which absorption and luminescence spectra can cover near-UV, visible and near-IR ranges of electromagnetic radiation. The luminophores of a new class have the high absorption coefficients (up to 200000 cm2/g) and the high luminescence quantum yields (up to 99%) with the decay times from 0.7 to 11 ns (Table 1). The good solubility of NOLs in organic solvents allows one to use solution techniques for production of different devices. Thus, a simple chemical modification of NOLs enables the production of new luminescent composite materials based on organosilicon polymers [71].
Table 1. Optical properties of NOLs
|
Cat. no |
Absorption |
K×104, |
Luminescence |
FWHM, nm |
PLQY, % |
PL decay |
Emission |
|
NOL1 |
213, 262, 342 |
8.0 |
390, 412 |
55 |
80 |
0.75 |
|
|
NOL2 |
213, 262, 366 |
6.7 |
390, 420 |
42 |
78 |
0.86 |
|
|
NOL3 |
213, 262, 335 |
5.8 |
377, 390 |
45 |
85 |
0.73 |
|
|
NOL4 |
213, 262, 367 |
7.4 |
396, 420 |
65 |
96 |
1.04 |
|
|
NOL5 |
213, 262, 375, 396 |
12.5 |
416, 436 |
60 |
82 |
4.47 |
|
|
NOL6 |
213, 262, 316, 457 |
6.5 |
588 |
120 |
87 |
7.75 |
|
|
NOL7 |
296, 367 |
9.6 |
396, 419 |
70 |
96 |
|
|
|
NOL8 |
337, 348 |
6.6 |
396, 419, 443 |
38 |
85 |
0.87 |
|
|
NOL9 |
327, 455 |
7.4 |
588 |
115 |
95 |
7.17 |
|
|
NOL10 |
337, 513 |
6.1 |
655 |
95 |
78 |
5.23 |
|
|
NOL11 |
332 |
11.7 |
396, 420 |
65 |
96 |
0.98 |
|
|
NOL12 |
296, 369 |
9.8 |
398, 422 |
65 |
98 |
0.98 |
|
|
NOL13 |
319, 400 |
7.1 |
502 |
105 |
83 |
4.70 |
|
|
NOL14 |
302, 402 |
10.1 |
502 |
110 |
90 |
5.38 |
|
|
NOL15 |
375, 455 |
7.5 |
588 |
110 |
65 |
6.55 |
|
|
NOL16 |
296, 386 |
12.7 |
486 |
70 |
99 |
5.45 |
|
|
NOL17 |
340, 385 |
7.7 |
438, 468 |
70 |
94 |
0.9 |
|
|
NOL18 |
328, 388 |
8.4 |
438, 468 |
70 |
93 |
0.83 |
|
|
NOL19 |
340, 385 |
6.4 |
438, 468 |
72 |
87 |
0.93 |
|
|
NOL20 |
213, 262, 388 |
7.5 |
468 |
73 |
84 |
|
|
|
NOL21 |
332 |
9.5 |
396, 420 |
68 |
93 |
0.93 |
|
|
NOL22 |
321, 333, 345 |
11.5 |
398, 422 |
66 |
98 |
0.91 |
|
|
NOL23 |
337, 348 |
7.3 |
396, 419, 443 |
44 |
74 |
|
|
|
NOL24 |
308, 368 |
6.9 |
398, 419, 443 |
44 |
85 |
|
|
|
NOL25 |
310, 342 |
9.0 |
390, 412 |
56 |
75 |
|
|
|
NOL26 |
330 |
10.0 |
392, 412 |
50 |
85 |
|
|
|
NOL27 |
333, 342 |
7.1 |
391, 412, 439 |
48 |
87 |
|
|
|
NOL28 |
333, 376, 396 |
6.0 |
417, 433 |
51 |
82 |
4.30 |
|
|
NOL29 |
265, 362, 514 |
7.2 |
655 |
103 |
67 |
5.29 |
|
|
NOL30 |
379, 512 |
7.1 |
655 |
104 |
75 |
5.18 |
|
|
NOL31 |
351, 495 |
5.6 |
621 |
104 |
80 |
6.86 |
|
|
NOL32 |
358, 429 |
6.0 |
597 |
114 |
60 |
2.62 |
|
|
NOL33 |
314, 404 |
4.7 |
524 |
115 |
95 |
4.36 |
|
|
NOL34 |
316, 455 |
7.9 |
592 |
154 |
99 |
10.61 |
|
|
NOL35 |
364,512 |
5.6 |
655 |
87 |
62 |
5.76 |
|
|
NOL36 |
322, 416 |
9.6 |
541 |
121 |
73 |
4,44 |
|
|
NOL37 |
277, 386 |
3.2 |
487 |
70 |
97 |
7,36 |
|
|
NOL38 |
382 |
11.6 |
431, 458 |
69 |
88 |
0.95 |
|
|
NOL39 |
325, 406 |
11.0 |
539 |
121 |
81 |
3.50 |
|
|
NOL40 |
348, 424 |
7.2 |
591 |
113 |
64 |
2.68 |
|
|
NOL41 |
395, 501 |
6.5 |
650 |
104 |
76 |
5.72 |
|
|
NOL42 |
369, 509 |
6.5 |
644 |
111 |
80 |
4.87 |
|
|
NOL43 |
295, 335 |
10.1 |
465 |
90 |
99 |
1.90 |
|
|
NOL44 |
299, 335 |
11.8 |
463 |
99 |
99 |
2.20 |
|
|
NOL45 |
254, 335 |
7.4 |
464 |
97 |
99 |
2.08 |
|
|
NOL46 |
252, 337 |
7.5 |
459 |
94 |
65 |
1.17 |
|
|
NOL47 |
320, 385 |
6.9 |
470 |
78 |
75 |
|
|
|
NOL48 |
256, 353 |
7.9 |
457 |
98 |
65 |
0.68 |
|
|
NOL49 |
365, 496 |
10.3 |
615 |
132 |
81 |
4.92 |
|
|
NOL50 |
264, 301, 413 |
18.9 |
612 |
218 |
60 |
|
|
A favorable combination of luminescence properties and physicochemical characteristics of NOLs promoted the development of a series of promising devices for wavelength shifting by the downshifting mechanism on their base.
Among the publications on spectral shifters based on organoelement luminophores, of note are two series of works that deal with germanium- and boron-containing luminophores. Oshita et al. [72] tried to create luminophores featuring an efficient intramolecular energy transfer utilizing the chemistry of germanium. Dithienogermole (DTG) was chosen as one of the chromophores, to which different chromophoric groups (fluorene, terthiophene, pyrene, and bifluorene) were attached. The systems of various structures were successfully synthesized, but the maximum achieved efficiencies of an intramolecular energy transfer reached only 47%, and the luminescence quantum yields reached only 32% [72].
A lot of systems with efficient FRET were synthesized based on BODIPY [73]. For example, Yilmaz et al. [74] obtained a compound bearing four donor BODIPY fragments and a perylenediimide (PDI) moiety as an acceptor. The energy transfer efficiency evaluated based on the quenching of BODIPY luminescence was 99%. Unfortunately, no data were provided on the luminescence quantum yield of the resulting compound, which makes controversial its classification as an efficient spectral shifter. Bozdemir et al. [75] synthesized a series of compounds bearing several chromophores based on BODIPY. Each of these chromophores separately exhibited the high luminescence quantum yield. However, their combination into a single system with efficient FRET (over 90% for any combination) led to a dramatic decay of PLQY to several percents. Hence, the main drawbacks of wavelength shifters based on BODIPY are the reduced luminescence quantum yields on passing from separate chromophores to the systems with efficient FRET.
3. Applications of wavelength shifters
3.1. High energy physics
To detect different types of radiation, scintillation materials are used that are able to emit photons of visible and ultraviolet ranges under action of ionizing radiations [76]. These materials include plastic scintillators that represent solid amorphous solutions of two types of organic luminophores (activators and spectral shifters) in a polymer matrix (Fig. 10a). Under action of an ionizing radiation, the polymer matrix is excited; then, the energy is transferred to the activator and is reemitted by the spectral shifter. In the classical plastic scintillators, the activator and shifter are located randomly in the polymer matrix at the distances which essentially exceed those able to provide an efficient nonradiative energy transfer. We suggested using NOLs as luminescent additives for plastic scintillators (Fig. 10b). The structures of NOLs allow one to combine both the activator and spectral shifter in a single molecule with efficient FRET [50, 77]. In this case, the energy of the polymeric matrix is absorbed by the donor fragments of NOL, and then an efficient intramolecular energy transfer and emission of the visible light by the acceptor fragment occur in the range of the maximum sensitivity of a photodetector.

Figure 10. Schematic representation of working principles of classical plastic scintillators with a radiative energy transfer between the activator and spectral shifter (a) and plastic scintillators based on NOLs with FRET between the activator and spectral shifter (b) [51].
This approach allowed for a substantial increase in the light yield of a plastic scintillator. The irradiation of samples with the thicknesses of 0.2 mm from a conventional polystyrene scintillator (Sc0) and a scintillator based on NOL1 (NSc1) with α-particles with the energy of 5.49 MeV led to 50% increase of the light stream (Fig. 11). Therefore, the new class of NOL spectral shifters can be successfully used in production of plastic scintillators.

Figure 11. Amplitude spectra of the standard (Sc0) and NOL-based (NSc1) plastic scintillators.
Besides 3D plastic scintillators, scintillating and wavelength-shifting fibers also find application in high energy physics [78]. The use of scintillating fibers enables the creation of rapid and relatively small detectors for elementary particles with different forms [79]. Polystyrene scintillating fibers include p-terphenyl (рТР) or 2,5-diphenyloxazole (РРО) as a primary luminescent additive in the content of several weight percents. For the efficient registration of scintillations by instrumental methods, a secondary additive is usually introduced, namely, a spectral shifter, for example, 1,4-bis(5-phenyloxazol-2-yl)benzene (РОРОР) [80]. The use of NOLs promoted the development of a new class of scintillating fibers that feature very short decay times and high light yields [81]. The investigations with scintillating fibers based on NOLs showed that the resulting samples feature decay times in the range of 1.2–1.3 ns, which is almost two times faster than the best standard fiber emitting blue light and six times faster than the best fiber with the green emission.
Undoped CsI crystals are one of the fastest inorganic scintillators with the decay time of about 30 ns. However, scintillation radiation of these crystals falls in the UV-range with the maximum at 320 nm, where the quantum efficiency of Hamamatsu S8664-1010 silicon avalanche photodiodes does not exceed 30%. Therefore, the wavelength-shifting plates (WLSs) bearing small amounts of NOL9, NOL10, and NOL14 were suggested and produced. These WSLs were placed between CsI crystals and a photodetector (Fig. 12), which afforded an increase in the efficiency of a cosmic-ray detector in 3 times [82].
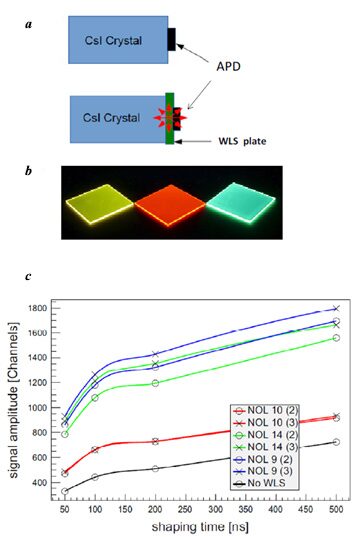
Figure 12. Schematic representation of pure CsI scintillator and APD photodetector connected directly or through a WLS plate (a). Photos of WLS plates based on NOL-9 (yellow), NOL-10 (red), and NOL-14 (green) (b). Signal from the counter with 2 S8664-1010 APDs and WLS plate (c) [82].
This effect was achieved owing to the fact that NOLs have the absorption maxima in the range of 300–330 nm and emit in the visible region of the spectrum with the maxima at 590, 650, and 500 nm in the case of NOL9, NOL10, and NOL14, respectively, where the quantum efficiency of APD composes about 85%.
Extensive use of organic spectral shifters in different devices for detection of elementary particles is caused by the low efficiency of different photodetectors in the ultraviolet spectral range. For example, in high energy physics, often different noble gases are used as media for detection of high-energy radiation and elementary particles [83]. However, they emit short-wave light: 127, 150, and 175 nm in the case of Ar, Kr, and Xe, respectively. Therefore, the detection is carried out using spectral shifters for reemitting of VUV photons to the visible light, where the sensitivity of cheep photodetectors is maximal. In particular, the application of tetraphenyl butadiene (TPB) [5, 25] and p-terphenyl [28] for this purpose was reported. Due to their polycrystalline structures, the matte coatings were obtained.
Akimov et al. [84] described the use of NOL1 and NOL3 as wavelength-shifting coatings for a scintillation detector based on liquid xenon. For this purpose, a sapphire window was applied with a thin layer of NOLs, which formed strong, transparent, amorphous films. NOLs in use possessed much better absorption in the region of xenon scintillation (175 nm) and emitted light (390 and 420 nm) which could be effectively registered by a photodetector (Fig. 13).

Figure 13. Emission spectrum of liquid Xe (1), absorption spectrum of p-terphenyl (2), absorption spectrum of NOL3 (3), absorptions spectrum of NOL1 (4), emission spectrum of p-terphenyl (5), emission spectrum of NOL3 (6), emission spectrum of NOL1 (7), photon detection efficiency (PDE) of the CPTA "blue-sensitive" photodiode (8), right axis. The absorption spectra of WLS shown in the units of optical density are normalized to the thickness of 200 nm. The emission spectra of WLS are normalized to 1 in arbitrary units [84].
Nowadays, there is a tendency to displace photodiodes in the devices for detection of elementary particles for silicon photomultipliers (SiPMs) [85–87]. Despite all the advantages, SiPMs are still poorly applicable for detection of VUV photons [88]. The coating of a conventional SiPM with a thin layer of NOL1 significantly enhanced its sensitivity in the ultraviolet region [89]. Figure 14 shows how the photodetection efficiency (PDE) of a SensLMicroFC-SMT-60035 silicon multiplier changed after the corresponding modification, which enhanced the photosensitivity in the UV region to 13–15%. This simple approach facilitates the use of conventional cheep SiPМs for detection of ultraviolet light.
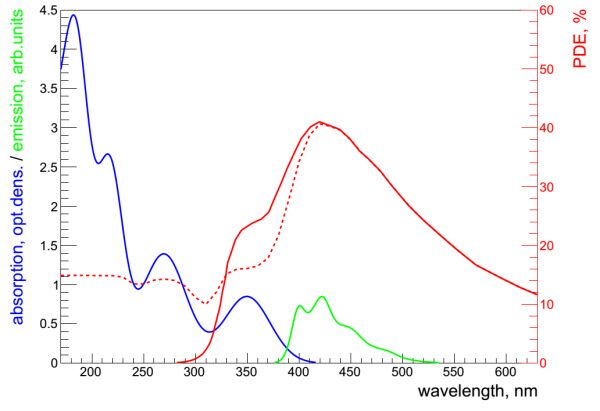
Figure 14. Absorption (blue) and luminescence (green) spectra of NOL1 and the spectral dependence of SiPM PDE without (red solid)
and with (red dashed) a 200 nm layer of NOL1 [89].
Hence, it was shown that new spectral shifters, exhibiting an efficient energy transfer, hold great promise for improving the efficiency of different detectors in high energy physics [90].
3.2. Luminescent downshifting for solar cells
The problem of creation of efficient renewable energy sources is one of the most pressing challenges for humanity. Photovoltaic cells convert solar energy to electrical energy; therefore, many studies aim at increasing their efficiency. One of the approaches to improve the efficiency factor of solar cells is the use of luminescent downshifting (LDS) layers [91]. They convert an incident short-wave radiation to a long-wave one, which can usually be utilized by photoelectric devices more effectively. For the first time, the concept of LDS layers was suggested in the 1970s in luminescent solar concentrators (LSCs) [92, 93]. Hovel et al. was the first to realize LDS experimentally by placing a photoluminescent material in the form of a wavelength-shifting plate in a solar cell [94].
Different classes of organic compounds were utilized in LDS layers [95–101]. Spectral shifters used in the production of LDS layers must combine good absorption in the range, where the conversion efficiency of a photovoltaic cell is low, with the low absorption and maximum luminescence in the region of the highest efficiency of a solar cell and simultaneously retain these properties as long as possible. This leads to the following requirements to luminophores: high luminescence quantum yield, high molar extinction coefficient, high Stokes shift, and high stability. These properties are characteristic, for example, for NOL15, which was introduced into different polymeric matrices for production of LDS layers. The resulting polymeric films were used for modification of copper–indium–gallium–(di)selenide (CIGS) solar cells, which exhibit low efficiency in the UV and blue spectral ranges (Fig. 15) [102]. The application of fine plates from a transparent polymer bearing a small additive of NOL15, which absorb photons in the range of 300–450 nm and reemit at 500–700 nm with the quantum yields from 80% to 92%, afforded an increase in the external quantum efficiency (EQE) of the device at 360 nm from 1% to 55%, which led to a relative increase in the short-circuit current and the efficiency of energy conversion by 3.2–4.3%.

Figure 15. EQE of a CIGS solar cell with an NOL-PVB layer [102].
3.3. Light-emitting diode sources of light
The problem of energy-conserving, efficient and safe illumination is topical for many people of all ages who spend the most parts of their lives indoor. A transition from incandescent light bulbs to modern light sources led to an essential reduction in the energy expenditures for illumination of working and living spaces. At the same time, the widespread introduction of illumination based on light-emitting diodes (LEDs) can significantly worsen the situation with eye diseases, especially among children and adolescents [103]. This can be connected with the presence of a large fraction of violet and blue light in the emission spectra of light-emitting diodes (Fig. 16). As early as the mid-1970s, it was shown that the detectable thresholds for retinal light damages in the blue spectral region (440–460 nm) are 50–100 times lower than those for the more long-wave light (500–700 nm) [104]. Retinal damages caused by a short-wave visible radiation occur due to a photochemical chain reaction, which consequences are accumulated during a whole life. This leads to accumulation of lipofuscin—a phototoxic aging pigment, which selectively absorbs light in the range of 440–460 nm and generates free radicals poisoning a retinal pigment epithelium [105]. Furthermore, the spectral characteristics of illumination strongly affect the production of melatonin—a hormone produced by the pineal gland, which is required for the normal operation of biological clocks and displays antioxidant and antitumor properties [106].
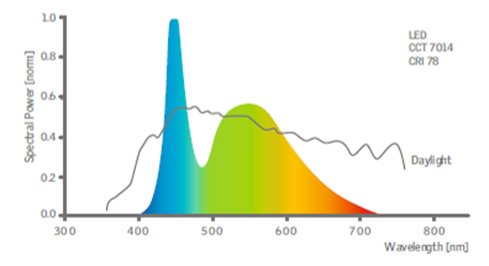
Figure 16. Spectral composition of a LED lamp.
Conventional light-emitting diode lamps are based on blue light-emitting diodes, which emit light with the wavelength of 460 nm, and luminophores, which convert it to yellow. In this case, white light represents a combination of blue and yellow lights with an essential blank in the range of 480–530 nm. Nowadays, there are two main approaches for construction of light-emitting diode illumination devices. The first one implies that the luminophore is located in the close proximity of the blue light source, forming the so-called white light-emitting diode. The luminophore is subjected to significant thermal impacts, which leads to accelerated degradation. Using this approach for creation of a light-emitting diode illumination lamp, it is necessary to join separate bright white light-emitting diodes in a single lighting device. This leads to nonuniform lighting and, as a result, to the blinding effect. The second approach is based on the application of a technology of coating of the luminophore with a single layer of a batch of low-power blue light-emitting diodes—the so-called remote luminophore concept. The realization of this concept implies that the luminophore is applied on a glass or is mixed with a polymer matrix, resulting in the formation of luminescent materials. It was shown that the application of the remote luminophore technology can improve the luminous efficiency and provide diffused light without a blinding effect [107].
The most popular light-emitting diode devices utilize blue light-emitting diodes with the wavelength of 450–460 nm and inorganic yellow luminophores based on Y3Al5O12:Ce3+ (YAG:Ce) [108]. However, most of oxide luminophores have low extinction coefficients in the visible part of the spectrum [109], i.e., they poorly absorb light from blue light-emitting diodes. Furthermore, they are prone to thermal quenching, which leads to changes in the diode coloration. Among organic compounds suggested for the use as wavelength-shifting materials for blue light-emitting diodes in white light devices, of note are polyfluorene derivatives [110]. Azide derivatives of polyfluorenes form cross-linked solid films, which have the absorption maxima at 387 nm, and, consequently, can be used only with the more expensive ultraviolet diodes. Zhang et al. suggested the use of triphenylamine derivatives, which emit yellow light, as donor-acceptor luminophores for white light-emitting diodes [111]. Recently, Kajjam et al. studied the effect of a ratio of donor and acceptor fragments in these systems on their luminescence properties [112]. They showed that an increase in the number of acceptor fragments from 1 to 3 leads to a displacement of the luminescence maximum in the solid state from 556 to 576, and further to 594 nm, respectively; but at the same time, the luminescence quantum yield reduces from 44% to 22%. The application of naphthalimide derivatives as luminophores distributed over an epoxide matrix to convert blue light of an InGaN light-emitting diode afforded pure white light [113]. However, the luminescence efficiency rapidly reduced in time. The authors explained this by the presence of water traces and monomer residues in the composite material, which can serve as proton sources that enter photochemical reactions with the naphthalimide derivatives. A substitution of the epoxide matrix for a poly(methyl methacrylate) polymer significantly increased the lifetime of such a light source. BODIPY derivatives were also used for light reemission from a blue light-emitting diode for production of white light [114].
Different NOLs were successfully used for correction of the spectral characteristics of light-emitting diode lamps. The application of NOLs ensured the creation of a biologically safe light source with very good characteristics and a light spectrum close to the solar one (Fig. 17). The color rendering index (CRI) of this lamp composes 95, and the efficiency reaches 123 lm/W.

Figure 17. Spectral composition of the LED lamp based on NOL (obtained by V. P. Gordienko, “Diodosvet” LLC, Voronezh, Russia).
Hence, it was shown that the application of NOLs can afford light-emitting lamps which combine the biologically safe light spectrum and the high energetic characteristics.
4. Conclusions and future outlook
In this review, we showed that efficient spectral shifters must possess the following properties: a broad absorption cross-section, which is related to the molar absorption coefficient (ε), the highest Stokes shift (S), and a high photoluminescence quantum yield (PLQY). The strategies for design and tuning of the optical properties and possible application fields of the novel efficient spectral shifters based on nanostructured organosilicon luminophores (NOLs), exhibiting intramolecular energy transfer by the Förster mechanism, were presented. The unique structures of NOLs allow for the variation of their optical properties over a broad spectral range. Furthermore, the maxima of absorption and emission spectra can be tuned independently from one another. It was shown that the new spectral shifters with efficient intramolecular energy transfer hold great promise for improvement of the efficiency of different detectors in high energy physics and the external quantum efficiency of photoelements, and may find application in creation of biologically safe light-emitting diode lamps. A catalog of LumInnoTech LLC [64] lists over 50 NOLs featuring various optical properties. We envisage that the further development of NOLs will proceed in the following three main directions: fundamental research on the processes of energy transfer, design and synthesis of new systems, and industrial implementation of the spectral shifters devised. Elucidation of the peculiarities of the mechanism of an intramolecular energy transfer will facilitate further creation of new systems with the desired optical properties. The structures of NOLs enable their functionalization with new reactive groups at the periphery. This offers new opportunities for creation of polymer composites, which production was limited earlier by the solubility of luminophores in a polymer matrix. The previous studies showed that the conjugation is not accomplished through positions 1, 3, and 5 of a benzene ring [115, 116]. Therefore, the substitution of a silicon atom for a benzene ring with different chromophores at positions 1, 3, and 5 can afford a new class of organic luminophores with an efficient intramolecular energy transfer. Widespread introduction of cheap light sources based on light-emitting diodes can lead to a significant impairment of vision of population, especially in the case of children and adolescents. The first examples of lamps with biologically relevant radiation spectra offer opportunities for the industrial implementation of NOLs for production of safe light sources.
Acknowledgements
This work was supported by the Russian Foundation for Basic Research, project no. 18-29-17006.
Absorption and luminescence spectral studies were performed with the financial support from the Ministry of Science and Higher Education of the Russian Federation using the equipment of Collaborative Access Center "Сenter for Polymer Research" of ISPM RAS.
References
- N. Surin, O. Borshchev, S. Ponomarenko, M. Skorotetcky, B. Lubsandorzhiev, N. Lubsandorzhiev, A. Pakhorukov, Nucl. Instrum. Methods Phys. Res., Sect. A, 2014, 766, 160–162. DOI: 10.1016/j.nima.2014.05.124
- K. Allada, Ch. Hurlbut, L. Ou, B. Schmookler, A. Shahinyan, B. Wojtsekhowski, Nucl. Instrum. Methods Phys. Res., Sect. A, 2015, 782, 87–91. DOI: 10.1016/j.nima.2015.02.012
- V. Chepel, H. Araújo, J. Instrum., 2013, 8, R04001. DOI: 10.1088/1748-0221/8/04/R04001
- D. Yu. Akimov, A. V. Akindinov, I. S. Alexandrov, V. A. Belov, O. V. Borshchev, A. A. Burenkov, M. V. Danilov, A. G. Kovalenko, Y. N. Luponosov, S. A. Ponomarenko, V. N. Stekhanov, N. M. Surin, S. A. Zav'yalov, M. Yu. Yablokov, Nucl. Instrum. Methods Phys. Res., Sect. A, 2012, 695, 403–406. DOI: 10.1016/j.nima.2011.12.036
- The ArDM Collaboration, V. Boccone, P. K. Lightfoot, K. Mavrokoridis, C. Regenfus, C. Amsler, A. Badertscher, A. Bueno, H. Cabrera, M. C. Carmona-Benitez, M. Daniel, E. J. Daw, U. Degunda, A. Dell'Antone, A. Gendotti, L. Epprecht, S. Horikawa, L. Kaufmann, L. Knecht, M. Laffranchi, C. Lazzaro, D. Lussi, J. Lozano, A. Marchionni, A. Melgarejo, P. Mijakowski, G. Natterer, S. Navas-Concha, P. Otyugova, M. de Prado, P. Przewlocki, F. Resnati, M. Robinson, J. Rochet, L. Romero, E. Rondio, A. Rubbia, N. J. C. Spooner, T. Strauss, J. Ulbricht, T. Viant, J. Instrum., 2009, 4, P06001. DOI: 10.1088/1748-0221/4/06/P06001
- H. J. Hovel, R. T. Hodgson, J. M. Woodall, Sol. Energy Mater., 1979, 2, 19–29. DOI: 10.1016/0165-1633(79)90027-3
- G. Maggioni, A. Campagnaro, S. Carturan, A. Quaranta, Sol. Energy Mater. Sol. Cells, 2013, 108, 27–37. DOI: 10.1016/j.solmat.2012.08.009
- L. Danos, T. Parel, T. Markvart, V. Barrioz, W. S. M. Brooks, S. J. C. Irvine, Sol. Energy Mater. Sol. Cells, 2012, 98, 486–490. DOI: 10.1016/j.solmat.2011.11.009
- B. S. Richards, Sol. Energy Mater. Sol. Cells, 2006, 90, 1189–1207. DOI: 10.1016/j.solmat.2005.07.001
- K. Bando, K. Sakano, Y. Noguchi, Y. Shimizu, J. Light Visual Environ., 1998, 22 (1), 2–5. DOI: 10.2150/jlve.22.1_2
- N. Hirosakia, R.-J. Xie, K. Kimoto, Appl. Phys. Lett., 2005, 86, 211905. DOI: 10.1063/1.1935027
- K. Sakuma, K. Omichi, N. Kimura, M. Ohashi, D. Tanaka, N. Hirosaki, Y. Yamamoto, R.-J. Xie, T. Suehiro, Opt. Lett., 2004, 29, 2001–2003. DOI: 10.1364/OL.29.002001
- K. D. Oskam, R. T. Wegh, H. Donker, E. V. D van Loef, A. Meijerink, J. Alloys Compd., 2000, 300–301, 421–425. DOI: 10.1016/S0925-8388(99)00755-0
- D. L. Dexter, J. Chem. Phys., 1953, 21, 836–850. DOI: 10.1063/1.1699044
- D. L. Dexter, Phys. Rev., 1957, 108, 630–633. DOI: 10.1103/PhysRev.108.630
- J. L. Sommerdijk, A. Bril, A.W. de Jager, J. Lumin., 1974, 8, 341–343. DOI: 10.1016/0022-2313(74)90006-4
- W. W. Piper, J. A. DeLuca, F. S. Ham, J. Lumin., 1974, 8, 344–348. DOI: 10.1016/0022-2313(74)90007-6
- J. C. Goldschmidt, S. Fischer, Adv. Opt. Mater., 2015, 3, 510–535. DOI: 10.1002/adom.201500024
- R. E. Halsted, E. F. Apple, J. S. Prener, Phys. Rev. Lett., 1959, 2, 420–421. DOI: 10.1103/PhysRevLett.2.420
- N. Bloembergen, Phys. Rev. Lett., 1959, 2, 84–85. DOI: 10.1103/PhysRevLett.2.84
- Y. Mita, Appl. Phys. Lett., 1981, 39, 587–589. DOI: 10.1063/1.92833
- L. Y. Ang, M. E. Lim, L. C. Ong, Y. Zhang, Nanomedicine, 2011, 6, 1273–1288. DOI: 10.2217/nnm.11.108
- M. Pope, C. E. Swenberg, Electronic Processes in Organic Crystals, Clarendon Press, New York, 1982.
- B. M. Krasovitskii, B. M. Bolotin, Organic Luminescent Materials, VCH Publ., NewYork, 1988.
- P. Benetti, C. Montanari, G. L. Raselli, M. Rossella, C. Vignoli, Nucl. Instrum. Methods Phys. Res., Sect. A, 2003, 505, 89–92. DOI: 10.1016/S0168-9002(03)01027-1
- G. Eigen, E. Lorenz, Nucl. Instrum. Methods, 1979, 166, 165–168. DOI: 10.1016/0029-554X(79)90569-X
- B. K. Lubsandorzhiev, Сh. Spiering, L. A. Kuz'michev, Instrum. Exp. Tech., 2002, 45, 742–746. DOI: 10.1023/A:1021462501792
- S. Belogurov, A. Bolozdynya, D. Churakov, A. Koutchenkov, V. Morgunov, V. Solovov, G. Safronov, G. Smirnov, V. Egorov, S. Medved, IEEE Nucl. Sci. Symp. Med. Imaging Conf. Rec., 1995, 1, 519–523. DOI: 10.1109/NSSMIC.1995.504280
- D. A. Pyatakov, O. V. Borshchev, M. S. Skorotetcky, S. A. Ponomarenko, Russ. J. Org. Chem., 2019, 55, 1619–1626. DOI: 10.1134/S051474921910015X
- V. A. Postnikov, N. I. Sorokina, O. A. Alekseeva, A. A. Kulishov, R. I. Sokolnikov, M. S. Lyasnikova, V. V. Grebenev, O. V. Borshchev, M. S. Skorotecky, N. M. Surin, E. A. Svidchenko, S. A. Ponomarenko, A. E. Voloshin, Crystallogr. Rep., 2018, 63, 819–831. DOI: 10.1134/S1063774518050243
- O. D. Parashchuk, A. A. Mannanov, V. G. Konstantinov, D. I. Dominskiy, N. M. Surin, O. V. Borshchev, S. A. Ponomarenko, M. S. Pshenichnikov, D. Yu. Paraschuk, Adv. Funct. Mater., 2018, 28, 1800116. DOI: 10.1002/adfm.201800116
- V. A. Postnikov, N. I. Sorokina, O. A. Alekseeva, A. A. Kulishov, R. I. Sokolnikov, M. S. Lyasnikova, V. V. Grebenev, O. V. Borshchev, M. S. Skorotecky, N. M. Surin, E. A. Svidchenko, S. A. Ponomarenko, A. E. Voloshin, Crystallogr. Rep., 2018, 63, 819–831. DOI: 10.1134/S1063774518050243
- V. A. Postnikov, M. S. Lyasnikova, A. A. Kulishov, V. V. Grebenev, O. V. Borshchev, Russ. J. Phys. Chem. A, 2019, 93, 1741–1746. DOI: 10.1134/S0036024419090176
- B. M. Krasovitskii, B. M. Bolotin, Chem. Heterocycl. Compd., 1974, 10, 1269–1284. DOI: 10.1007/BF01175078
- M. S. Skorotetcky, E. D. Krivtsova, O. V. Borshchev, N. M. Surin, E. A. Svidchenko, Y. V. Fedorov, S. A. Pisarev, S. A. Ponomarenko, Dyes Pigm., 2018, 155, 284–291. DOI: 10.1016/j.dyepig.2018.03.043
- C. Leroy, P.-G. Rancoita, Principles of Radiation Interaction in Matter and Detection, 3rd Edition, World Sci., Singapore, 2011.
- V. A. Postnikov, Y. I. Odarchenko, A. V. Iovlev, V. V. Bruevich, A. Yu. Pereverzev, L. G. Kudryashova, V. V. Sobornov, L. Vidal, D. Chernyshov, Y. N. Luponosov, O. V. Borshchev, N. M. Surin, S. A. Ponomarenko, D. A. Ivanov, D. Yu. Paraschuk, Cryst. Growth Des., 2014, 14, 1726–1737. DOI: 10.1021/cg401876a
- L. G. Kudryashova, M. S. Kazantsev, V. A. Postnikov V. V. Bruevich, Yu. N. Luponosov, N. M. Surin, O. V. Borshchev, S. A. Ponomarenko, M. S. Pshenichnikov, D. Yu. Paraschuk, ACS Appl. Mater. Interfaces, 2016, 8, 10088–10092. DOI: 10.1021/acsami.5b11967
- Phosphor Handbook, 2nd Edition, S. Shionoya, W. M. Yen, H. Yamamoto (Eds.), CRC Press, Boca Raton, 2018.
- A. Yu. Sosorev, M. K. Nuraliev, E. V. Feldman, D. R. Maslennikov, O. V. Borshchev, M. S. Skorotetcky, N. M. Surin, M. S. Kazantsev, S. A. Ponomarenko, D. Yu. Paraschuk, Phys. Chem. Chem. Phys., 2019, 21, 11578–11588. DOI: 10.1039/C9CP00910H
- M. S. Skorotetcky, O. V. Borshchev, N. M. Surin, Y. Odarchenko, S. A. Pisarev, S. M. Peregudova, K. W. Törnroos, D. Chernyshov, D. A. Ivanov, S. A. Ponomarenko, Dyes Pigm., 2017, 141, 128–136. DOI: 10.1016/j.dyepig.2017.02.006
- H.-J. Kim, J.-Y. Jin, Y.-S. Lee, S.-H. Lee, C.-H. Hong, Chem. Phys. Lett., 2006, 431, 341–345. DOI: 10.1016/j.cplett.2006.09.090
- A. Loudet, K. Burgess, Chem. Rev., 2007, 107, 4891–4932. DOI: 10.1021/cr078381n
- N. Boens, V. Leen, W. Dehaen, Chem. Soc. Rev., 2012, 41, 1130–1172. DOI: 10.1039/C1CS15132K
- A. Bessette, G. S. Hanan, Chem. Soc. Rev., 2014, 43, 3342–3405. DOI: 10.1039/C3CS60411J
- H. Lu, J. Mack, Y. Yang, Z. Shen, Chem. Soc. Rev., 2014, 43, 4778–4823. DOI: 10.1039/C4CS00030G
- A. A. Pakhomov, Y. N. Kononevich, M. V. Stukalova, E. A. Svidchenko, N. M. Surin, G. V. Cherkaev, O. I. Shchegolikhina, V. I. Martynov, A. M. Muzafarov, Tetrahedron Lett., 2016, 57, 979–982. DOI: 10.1016/j.tetlet.2016.01.059
- L. Jiao, C. Yu, T. Uppal, M. Liu, Y. Li, Y. Zhou, E. Hao, X. Hu, M. G. H. Vicente, Org. Biomol. Chem., 2010, 8, 2517–2519. DOI: 10.1039/C001068E
- N. J. Findlay, J. Bruckbauer, A. R. Inigo, B. Breig, S. Arumugam, D. J. Wallis, R. W. Martin, P. J. Skabara, Adv. Mater., 2014, 26, 7290–7294. DOI: 10.1002/adma.201402661
- S. A. Ponomarenko, N. M. Surin, O. V. Borshchev, Y. N. Luponosov, D. Y. Akimov, I. S. Alexandrov, A. A. Burenkov, A. G. Kovalenko, V. N. Stekhanov, E. A. Kleymyuk, O. T. Gritsenko, G. V. Cherkaev, A. S. Kechek'yan, O. A. Serenko, A. M. Muzafarov, Sci. Rep., 2014, 4, 6549. DOI: 10.1038/srep06549
- S. A. Ponomarenko, N. M. Surin, O. V. Borshchev, M. S. Skorotetcky, A. M. Muzafarov, Proc. SPIE, 2015, 9545, 954509. DOI: 10.1117/12.2187281
- O. V Borshchev, E. A. Kleymyuk, N. M. Surin, E. A. Svidchenko, Yu. V. Fedorov, P. V. Dmitryakov, S. N. Chvalun, S. A. Ponomarenko, Org. Photonics Photovoltaics, 2017, 5, 1–8. DOI: 10.1515/oph-2017-0001
- Y. N. Luponosov, S. A. Ponomarenko, N. M. Surin, O. V. Borshchev, E. A. Shumilkina, A. M. Muzafarov, Chem. Mater., 2009, 21, 447–455. DOI: 10.1021/cm8022488
- O. V. Borshchev, S. A. Ponomarenko, E. A. Kleymyuk, Y. N. Luponosov, N. M. Surin, A. M. Muzafarov, Russ. Chem. Bull., 2010, 59, 797–805. DOI: 10.1007/s11172-010-0164-6
- C. Devadoss, P. Bharathi, J. S. Moore, J. Am. Chem. Soc., 1996, 118, 9635–9644. DOI: 10.1021/ja961418t
- Th. Förster, Discuss. Faraday Soc., 1959, 27, 7–17. DOI: 10.1039/DF9592700007
- T. Yu. Starikova, N. M. Surin, O. V. Borshchev, S. A. Pisarev, E. A. Svidchenko, Yu. V. Fedorov, S. A. Ponomarenko, J. Mater. Chem. C, 2016, 4, 4699–4708. DOI: 10.1039/c6tc00979d
- M. S. Polinskaya, O. V. Borshchev, Y. N. Luponosov, N. M. Surin, A. M. Muzafarov, S. A. Ponomarenko, Mendeleev Commun., 2011, 21, 89–91. DOI: 10.1016/j.mencom.2011.03.010
- N. M. Surin, O. V. Borshchev, Yu. N. Luponosov, S. A. Ponomarenko, A. M. Muzafarov, Russ. J. Phys. Chem. A, 2010, 84, 1979–1985. DOI: 10.1134/S0036024410110270
- M. S. Skorotetcky, O. V. Borshchev, G. V. Cherkaev, S. A. Ponomarenko, Russ. J. Org. Chem., 2019, 55, 25–41. DOI: 10.1134/S1070428019010056
- S. A. Ponomarenko, A. M. Muzafarov, O. V. Borshchev, E. A. Vodopyanov, N. V. Demchenko, V. D. Myakushev, Russ. Chem. Bull., 2005, 54, 684–690. DOI: 10.1007/s11172-005-0306-4
- O. V. Borshchev, S. A. Ponomarenko, N. M. Surin, M. M. Kaptyug, M. I. Buzin, A. P. Pleshkova, N. V. Demchenko, V. V. Myakushev, A. M. Muzafarov, Organometallics, 2007, 26, 5165–5173. DOI: 10.1021/om0702328
- V. P. Ananikov, L. L. Khemchyan, Yu. V. Ivanova, V. I. Bukhtiyarov, A. M. Sorokin, I. P. Prosvirin, S. Z. Vatsadze, A. V. Medved'ko, V. N. Nuriev, A. D. Dilman, V. V. Levin, I. V. Koptyug, K. V. Kovtunov, V. V. Zhivonitko, V. A. Likholobov, A. V. Romanenko, P. A. Simonov, V. G. Nenajdenko, O. I. Shmatova, V. M. Muzalevskiy, M. S. Nechaev, A. F. Asachenko, O. S. Morozov, P. B. Dzhevakov, S. N. Osipov, D. V. Vorobyeva, M. A. Topchiy, M. A. Zotova, S. A. Ponomarenko, O. V. Borshchev, Yu. N. Luponosov, A. A. Rempel, A. A. Valeeva, A. Yu. Stakheev, O. V. Turova, I. S. Mashkovsky, S. V. Sysolyatin, V. V. Malykhin, G. A. Bukhtiyarova, A. O. Terent'ev, I. B. Krylov, Russ. Chem. Rev., 2014, 83, 885–985. DOI: 10.1070/RC2014v83n10ABEH004471
- www.luminnotech.com
- RU Patent 2544863 С2, 2015.
- RU Patent 2524960 C1, 2014.
- EP Patent 3006486 B1, 2018.
- US Patent 9777026 B2, 2017.
- US Patent 10131678 B2, 2018.
- RU Patent 2620088 C1.
- M. S. Skorotetcky, O. V. Borshchev, N. M. Surin, I. B. Meshkov, A. M. Muzafarov, S. A. Ponomarenko, Silicon, 2015, 7, 191–200. DOI: 10.1007/s12633-014-9256-5
- Y. Adachi, T. Nomura, J. Ohshita, Chem. Eur. J., 2019, 25, 4974–4983. DOI: 10.1002/chem.201805418
- R. Ziessel, A. Harriman, Chem. Commun., 2011, 47, 611–631. DOI: 10.1039/c0cc02687e
- M. D. Yilmaz, O. A. Bozdemir, E. U. Akkaya, Org. Lett., 2006, 8, 2871–2873. DOI: 10.1021/ol061110z
- O. A. Bozdemir, S. Erbas-Cakmak, O. O. Ekiz, A. Dana, E. U. Akkaya, Angew. Chem., Int. Ed., 2011, 50, 10907–10912. DOI: 10.1002/anie.201104846
- A. Peurrung, Mater. Today, 2008, 11, 50–54. DOI: 10.1016/S1369-7021(08)70019-1
- M. S. Skorotetcky, O. V. Borshchev, N. M. Surin, S. A. Ponomarenko, J. Phys.: Conf. Ser., 2018, 1124, 051010. DOI: 10.1088/1742-6596/1124/5/051010
- R. C. Ruchti, Annu. Rev. Nucl. Part. Sci., 1996, 46, 281–319. DOI: 10.1146/annurev.nucl.46.1.281
- C. Joram, G. Haefeli, B. Leverington, J. Instrum., 2015, 10, C08005. DOI: 10.1088/1748-0221/10/08/C08005
- F. Takasaki, H. Saito, T. Shimizu, S. Kondo, O. Shinji, Nucl. Instrum. Methods Phys. Res., Sect. A, 1987, 262, 224–228. DOI: 10.1016/0168-9002(87)90860-6
- O. Borshchev, A. B. R. Cavalcante, L. Gavardi, L. Gruber, C. Joram, S. Ponomarenko, O. Shinji, N. Surin, J. Instrum., 2017, 12, P05013. DOI: 10.1088/1748-0221/12/05/P05013
- Y. Jin, H. Aihara, O. V. Borshchev, D. A. Epifanov, S. A. Ponomarenko, N. M. Surin, Nucl. Instrum. Methods Phys. Res., Sect. A, 2016, 824, 691–692. DOI: 10.1016/j.nima.2015.07.034
- V. Chepel, H. Araújo, J. Instrum., 2013, 8, R04001. DOI: 10.1088/1748-0221/8/04/R04001
- D. Yu. Akimov, A. V. Akindinov, I. S. Alexandrov, V. A. Belov, O. V. Borshchev, A. A. Burenkov, M. V. Danilov, A. G. Kovalenko, Y. N. Luponosov, S. A. Ponomarenko, V. N. Stekhanov, N. M. Surin, S. A. Zav'yalov, M. Yu. Yablokov, Nucl. Instrum. Methods Phys. Res., Sect. A, 2012, 695, 403–406. DOI: 10.1016/j.nima.2011.12.036
- T. Kirn, Nucl. Instrum. Methods Phys. Res., Sect. A, 2017, 845, 481–485. DOI: 10.1016/j.nima.2016.06.057
- N. Strobbe, J. Instrum., 2017, 12, C01080. DOI: 10.1088/1748-0221/12/01/C01080
- S Ogawa, Nucl. Instrum. Methods Phys. Res., Sect. A, 2017, 845, 528–532. DOI: 10.1016/j.nima.2016.06.085
- D. Renker, E. Lorenz, J. Instrum., 2009, 4, P04004. DOI: 10.1088/1748-0221/4/04/P04004
- D. Yu. Akimov, V. A. Belov, O. V. Borshchev, A. A. Burenkov, Yu. L. Grishkin, A. K. Karelin, A. V. Kuchenkov, A. N. Martemiyanov, S. A. Ponomarenko, G. E. Simakov, V. N. Stekhanov, N. M. Surin, V. S. Timoshin, O. Ya. Zeldovich, J. Instrum., 2017, 12, P05014. DOI: 10.1088/1748-0221/12/05/P05014
- S. A. Ponomarenko, O. V. Borshchev, N. M. Surin, M. S. Skorotetcky, E. A. Kleymyuk, T. Yu. Starikova, A. S. Tereshenko, Proc. SPIE, 2017, 10344, 103440N. DOI: 10.1117/12.2273981
- E. Klampaftis, D. Ross, K. R. McIntosh, B. S. Richards, Sol. Energy Mater. Sol. Cells, 2009, 93, 1182–1194. DOI: 10.1016/j.solmat.2009.02.020
- A. Goetzberger, W. Greube, Appl. Phys., 1977, 14, 123–139. DOI: 10.1007/BF00883080
- W. H. Weber, J. Lambe, Appl. Opt., 1976, 15, 2299–2300. DOI: 10.1364/AO.15.002299
- H. J. Hovel, R. T. Hodgson, J. M. Woodall, Sol. Energy Mater., 1979, 2, 19–29. DOI: 10.1016/0165-1633(79)90027-3
- G. Maggioni, A. Campagnaro, S. Carturan, A. Quaranta, Sol. Energy Mater. Sol. Cells, 2013, 108, 27–37. DOI: 10.1016/j.solmat.2012.08.009
- L. Danos, T. Parel, T. Markvart, V. Barrioz, W. S. M. Brooks, S. J. C. Irvine, Sol. Energy Mater. Sol. Cells, 2012, 98, 486–490. DOI: 10.1016/j.solmat.2011.11.009
- A. Bessette, G. S. Hanan, Chem. Soc. Rev., 2014, 43, 3342–3405. DOI: 10.1039/C3CS60411J
- T. Maruyama, A. Enomoto, K. Shirasawa, Sol. Energy Mater. Sol. Cells, 2000, 64, 269–278. DOI: 10.1016/S0927-0248(00)00227-0
- L. H. Slooff, R. Kinderman, A. R. Burgers, N. J. Bakker, J. A. M. van Roosmalen, A. Büchtemann, R. Danz, M. Schleusener, J. Sol. Energy Eng., 2007, 129, 272–276. DOI: 10.1115/1.2735347
- T. Maruyama, Y. Shinyashiki, S. Osako, Sol. Energy Mater. Sol. Cells, 1998, 56, 1–6. DOI: 10.1016/S0927-0248(98)00105-6
- K. R. McIntosh, G. Lau, J. N. Cotsell, K. Hanton, D. L. Bätzner, F. Bettiol, B. S. Richards, Prog. Photovoltaics, 2009, 17, 191–197. DOI: 10.1002/pip.867
- T. Uekert, A. Solodovnyk, S. Ponomarenko, A. Osvet, I. Levchuk, J. Gast, M. Batentschuk, K. Forberich, E. Stern, H.-J. Egelhaaf, C. J. Brabec, Sol. Energy Mater. Sol. Cells, 2016, 155, 1–8. DOI: 10.1016/j.solmat.2016.04.019
- P. P. Zak, M. A. Ostrovsky, Light Eng., 2012, 20, 5–8.
- W. T. Ham, H. A. Mueller, D. H. Sliney, Nature, 1976, 260, 153–155. DOI: 10.1038/260153a0
- M. A. Yakovleva, T. B. Feldman, Z. M. Polonskaia, A. E. Dontsov, S. A. Borzenok, Kh. P. Takhchidi, M. A. Ostrovsky Fyodorov, J. Ophthalmic Surg., 2009, 5, 59–64.
- F. Falchi, P. Cinzano, C. D. Elvidge, D. M. Keith, A. Haim, J. Environ. Manage., 2011, 92, 2714–2722. DOI: 10.1016/j.jenvman.2011.06.029
- N. Narendran, Y. Gu, J. P. Freyssinier-Nova, Y. Zhu, Phys. Status Solidi A, 2005, 202, R60–R62. DOI: 10.1002/pssa.200510015
- S. Nakamura, G. Fasol, The Blue Laser Diode: GaN Based Light Emitters and Lasers, Springer, Berlin, 1996, pp. 216–221.
- R.-J. Xie, N. Hirosaki, Sci. Technol. Adv. Mater., 2007, 8, 588–600. DOI: 10.1016/j.stam.2007.08.005
- I. O. Huyal, U. Koldemir, T. Ozel, H. V. Demir, D. Tuncel, J. Mater. Chem., 2008, 18, 3568–3574. DOI: 10.1039/B802910E
- L. Zhang, B. Li, B. Lei, Z. Hong, W. Li, J. Lumin., 2008, 128, 67–73. DOI: 10.1016/j.jlumin.2007.05.009
- A. B. Kajjam, S. Giri, V. Sivakumar, Mater. Chem. Front., 2017, 1, 512–520. DOI: 10.1039/C6QM00031B
- H.-J. Kim, J.-Y. Jin, Y.-S. Lee, S.-H. Lee, C.-H. Hong, Chem. Phys. Lett., 2006, 431, 341–345. DOI: 10.1016/j.cplett.2006.09.090
- N. J. Findlay, J. Bruckbauer, A. R. Inigo, B. Breig, S. Arumugam, D. J. Wallis, R. W. Martin, P. J. Skabara, Adv. Mater., 2014, 26, 7290–7294. DOI: 10.1002/adma.201402661
- S. A. Ponomarenko, S. Kirchmeyer, A. Elschner, B.-H. Huisman, A. Karbach, D. Drechsler, Adv. Funct. Mater., 2003, 13, 591–596. DOI: 10.1002/adfm.200304363
- M. H. van der Veen, M. T. Rispens, H. T. Jonkman, J. C. Hummelen, Adv. Funct. Mater., 2004, 14, 215–223. DOI: 10.1002/adfm.200305003 DOI: 10.1002/adfm.200305003





